Novel monoclonal antibody development in just 40-60 days! Precision Antibody uses its proprietary procedures and platform to rapidly generate application specific monoclonal antibodies (mAbs). Precision Antibody customizes project strategies to ensure the delivery of mAbs that work in the intended applications.

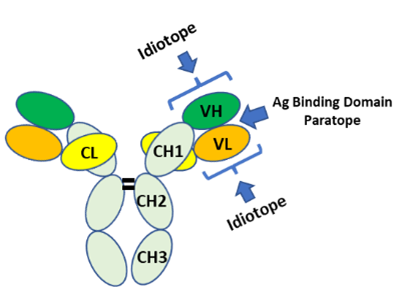
Precision Antibody has extensive expertise in developing anti-idiotypic antibodies, which specifically target the variable regions of drug antibodies (DAs). These monoclonal antibodies can identify DAs in biological fluids containing large amounts of endogenous antibodies, making them ideal for pharmacokinetic (PK) studies to monitor circulating DAs.
Anti-idiotype antibodies are categorized into two types: anti-paratope (competes with the antigen for binding to DA) and anti-idiotope (does not interfere with antigen binding). When generating anti-idiotype antibodies, it’s essential to differentiate between anti-paratope and anti-idiotope antibodies. Anti-paratope antibodies are unable to bind to antigen-bound DA, making them unsuitable for detecting total circulating DA.
Our clients typically request antibody pairs for developing sandwich ELISAs to measure circulating DAs. Precision Antibody selects high-affinity, low-off-rate capture and detection antibody pairs with minimal matrix effects (serum interference) for optimal PK study performance.

PA offers fully human antibody development using transgenic mice. Precision Antibody uses its proprietary procedures and platform to rapidly generate specific therapeutic monoclonal antibodies. We are capable of developing a large number of antigen-specific clones producing fully human IgG in about 60 days. Please inquire.
At times you may encounter the situation “I needed that antibody, yesterday!!” Well, PA cannot reverse the time to accommodate your needs, but we offer the next best option and develop high-affinity hyperimmune serum in less than 3 weeks using mice or rats. Affinity purification will add an additional 7-10 days. The development of rat polyclonal antibodies is the most popular option for many of our customers. Due to the high specific IgG titer we achieve, even a smaller amount of serum goes a long way in your research.
PA offers traditional rabbit polyclonal antibody development services. We have 60 or 90-day protocols and continuous production of antibodies is possible. We also offer antibody purification services from rabbit polyclonal serum. (see Antibody Production and Purification section)
Please contact our specialist for rabbit polyclonal service.
For immunization of larger animals, please inquire.
Experience a dedicated approach to custom antibodies with unmatched expertise, fast delivery, and personalized service. Let’s collaborate to bring your research to life.


Precision Antibody™ is the forefront of the global Custom Antibody industry & it is led by the innovative minds in immunology and antibody development field.